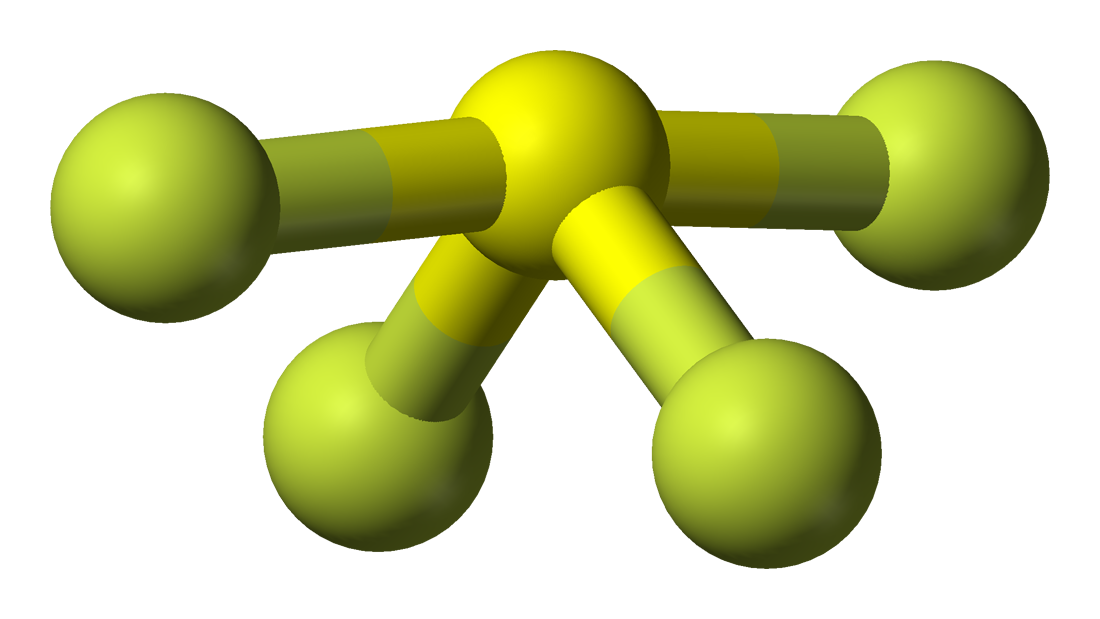
Choose The Best Lewis Structure For Sf4Choose the best Lewis structure for SF4. So, that bends the axial fluorines together a bit. molecular geometry and number of electron groups for SF4 seesaw 5 groups. Note though that the structure is distorted a bit due to the repulsive forces of the lone pair of electrons you see (not bonded). With one lone pair of valence electrons, you get a seesaw molecular geometry. The molecular geometry of SF4 is seesaw with an asymmetric distribution of electrons around the central atom. 
The seesaw shape of this molecule is due to the lone pairs of electrons. 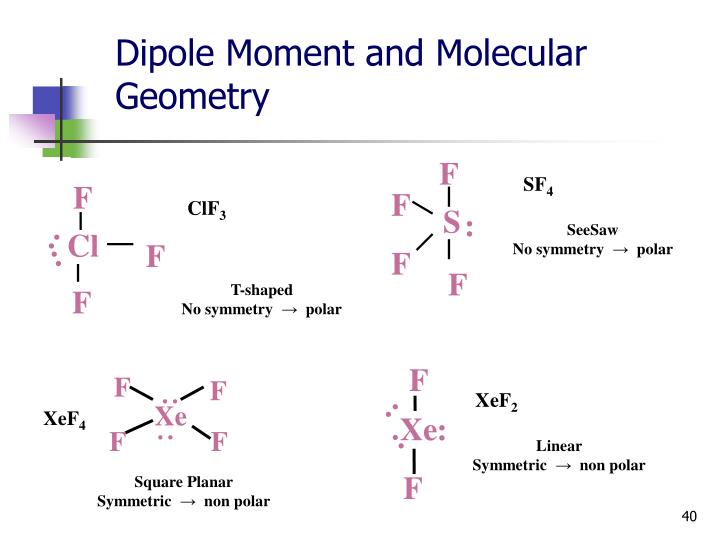
To identify this, use the Lewis Structure. Therefore, you can put 6x4 on each fluorine, 2x4 to account for four single bonds, and 2 for the last 2 valence electrons available.Īs a result, you have 5 electron groups, so the electron geometry would be trigonal bipyramidal. The first element that can identify if the Sulfur Tetrafluoride is polar is by its molecular geometry. You can put sulfur in the middle because fluorine tends to make single bonds. The three fluorine atoms and the two lone pairs of electrons on the sulfur atom occupy these regions, resulting in a trigonal bipyramidal shape. This means that the sulfur atom is surrounded by five regions of electron density. B) 0 lone pairs, trigonal bipyramidal E) 2 lone pairs, pentagonal. The molecular geometry of SF4 is trigonal bipyramidal. In the Lewis Structure, the unsymmetrical seesaw structure of SF4 leads to an overall dipole. A) 0 lone pairs, square pyramidal D) 1 lone pair, square pyramidal. Within the context of VSEPR theory, you can count electrons to determine the electron geometry ("parent" geometry). Give the number of lone pairs around the central atom and the molecular geometry of IF5.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
